| Case Report | ||
J. Microbiol. Infect. Dis., (2023), Vol. 13(2): 87–89 Case Report A case of melioidosis with hemoptysis: Strategies to isolate and identify Burkholderia pseudomalleiTherese Mary Dhason*, Thyagarajan Ravinder, Usha Krishnan, Pavithra Ammanarasimman, Lavanya T. Kamalasekaran and Amutha ChellaiahGovernment Kilpauk Medical College, Chennai, India *Corresponding Author: Therese Mary Dhason. Government Kilpauk Medical College, Chennai, India. Email: tmary1964 [at] gmail.com Submitted: 14/11/2022 Accepted: 04/06/2023 Published: 30/06/2023 © 2023 Journal of Microbiology and Infectious Diseases
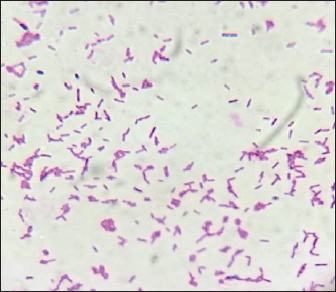
AbstractBackground: Melioidosis is an infectious disease caused by Burkholderia pseudomallei with diverse clinical manifestations. Mortality is due to septic shock and pneumonia. Burkholderia pseudomallei is a Gram-negative rod with bipolar staining. Culture is the gold standard diagnostic test. Gram stain, biochemical reactions, and Poymyxin resistance aid in the identification of the organism. Case Description: A 52-year-old female with type 2 diabetes mellitus was hospitalized for a chronic cough. A sputum sample was collected for Culture and Acidfast staining. Burkholderia pseudomallei was isolated and the patient was prescribed appropriate antibiotic. On the 15th day of admission, patient expired following a bout of hemoptysis despite antibiotic therapy. Conclusion: Culture is the gold standard diagnostic method in melioidosis. Burholderia pseudomallei can be easily differentiated from other organisms by Gram stain, methylene blue stain, and screening for polymyxin sensitivity. Early diagnosis and appropriate antibiotic therapy are warranted in melioidosis. Keywords: Burkholderia pseudomallei, Bipolar staining, Polymyxin B. IntroductionMelioidosis is an infectious disease that presents either as an acute or chronic infection. The causative organism is Burkholderia pseudomallei. In humans’ infection occurs by contact with contaminated soil and water. The incubation period is 1–21 days. The symptoms of melioidosis are fever, skin lesions, cough, breathlessness, and hemoptysis. Pneumonia, abscess, and sepsis are the various presentations of the disease. Septic shock is one of the causes of mortality (Wiersinga et al., 2018; Fertitta et al., 2019). In 10% of the individuals symptoms persist for more than 2 months leading to chronic melioidosis which mimics tuberculosis (TB) (Foong et al., 2014). Isolation of B. pseudomallei from specimens such as sputum, throat swab, pus from abscesses by culture has a sensitivity of 60% in the diagnosis of melioidosis (Limmathurotsakul et al., 2010). It grows on any blood agar medium, MacConkey agar, and Ashdown’s medium which contains antibiotics like gentamycin or colistin (Currie, 2015). The culture plates should be incubated at 37°C and should be observed for growth for 4 days. The colonies are creamy and non-hemolytic initially and become dry and wrinkled on the 4th day of incubation. Gram stain, Methylene blue stain, Motility test biochemical tests, and screening for polymyxin B sensitivity are the tests of identification of B. pseudomallei. Antigen detection tests like latex agglutination test and ELISA test are other diagnostic tests for melioidosis (Suttisunhakul et al., 2016). Imaging techniques like X-ray chest and CT scan play a major role in the diagnosis of melioidosis. Microscopy reveals Gram-negative rods with bipolar staining resembling a closed safety pin (Klietmann and Ruoff, 2001). Treatment of melioidosis is by antibiotics which are sensitive to antibiotic susceptibility test. The drugs which commonly show sensitivity are ceftazidime, meropenem, imipenem, and co-amoxiclav. Polymyxin B is intrinsically resistant. Other antibiotics susceptible in B. pseudomallei are doxycycline, chloramphenicol, and co-trimoxazole. Though it is resistant to Gentamycin, in Malaysia 86% of the isolates are sensitive to Gentamycin (Lipsitz et al., 2012). Case DetailsA 52 years old female a known diabetic for 10 years was admitted in the internal medicine ward with a C/O cough for 3 months. The patient was investigated for pulmonary TB by acid fast staining of sputum and cartridge-based nucleic acid amplification test and was found to be TB negative. CT scan of the thorax showed bilateral cavities. Physicians suspected melioidosis as it mimics TB and the presentation was a chronic cough. Melioidosis is either under diagnosed or misdiagnosed as TB since the clinical features are somewhat similar in both infections (Vidyalakshmi et al., 2008). Patient was started on piperacillin-tazobactum and continued to receive it for 10 days. Sputum was received in our microbiology laboratory for culture and sensitivity after 10 days of antibiotic therapy and it was processed. Suddenly patient had a massive bout of haemoptysis on the 15th day of admission and expired. Culture showed lactose non-fermenting colonies on MacConkey agar, Gram-negative rods with bipolar staining on Gram stain (Fig. 1), bipolar stained rods on Methylene blue stain (Fig. 2) and was motile. Biochemical tests for identification of organism were done. It was oxidase positive, oxidative in O/F test, indole negative, K/K in triple sugar iron agar medium, citrate was utilized and urea was not hydrolyzed. Screening for Polymyxin B (300 U) resistance was done and it was found to be resistant.
Fig. 1. Gram stain showing bipolar staining (Closed safety pin appearance).
Fig. 2. Methylene blue stain showing bipolar staining. The organism was sensitive to Amoxycillin-clavulanic acid, Co-trimoxazole, Meropenem and resistant to Ampicilin, 3rdgeneration Cephalosporins, Aminoglycosides, Piperacillin-tazobactum and Chloramphenicol by Kirby-Bauer disc diffusion method. DiscussionFor the physicians it is a challenge to diagnose melioidosis is a challenge to diagnose as it mimics pulmonary TB clinically. From the microbiologists’ perspectives it has to be differentiated from Pseudomonas as the colonies are nonfermenters on MacConkey agar, oxidase positive, Oxidative in O/F test and resemble Pseudomonas in other biochemical reactions like indole test, TSI reaction and citrate utilization test (Doker et al., 2014; Lau et al., 2015; Wiersinga et al., 2018). Culture and isolation of the organism is the gold standard method in the diagnosis of melioidosis (Benoit et al., 2015). On MacConkey agar the colonies were round, translucent and lactose non fermenting and were non hemolytic on blood agar. Gram stain plays a major role in differentiating B. pseudomallei from Pseudomonas. Burholderia pseudomallei are Gram-negative rods with bipolar staining whereas Pseudomonas spp do not have bipolar staining. The organism was resistant to Polymyxin-B (300 U disc) which is one of the screening methods for identification of B. pseudomallei. A bench test to identify the organism is 3 antibiotic discs test using Gentamycin, Colistin, and Amoxycillin-clavulanic acid in which B. pseudomallei is sensitive to Amoxycillin-clavulanic acid (≥18 mm surrounding a 20/10 mg amoxicillin-clavulanic acid disk) and resistant to other two antibiotics (Trinh et al., 2018). In contrast, Pseudomonas is resistant to Amoxycillin-clavulanic acid and sensitive to Polymyxin B (Hodgson et al., 2009). Patient was given empirical treatment with the antibiotic piperacillin-tazobactam on admission. Once the culture and sensitivity report was ready patient was prescribed parenteral antibiotic Meropenem in therapeutic dosage. Burholderia pseudomallei has become resistant to various resistance groups of antibiotics like third-generation cephalosporins, penicillins, rifamycins, macrolides, quinolones, and aminoglycosides. Carbapenems are sensitive even in extended spectrum beta-lactamases. Pneumonia culminating in sepsis related complications is attributable to higher mortality despite appropriate antibiotic therapy. Therefore, early diagnosis is warranted in melioidosis ConclusionMelioidosis was described as glanders -like disease in 1911 from Rangoon, Burma in a drug addict (Whitmore, 1913). It is emerged as an infectious disease of public health concern in the later period of 20th century in Southeast Asia and Australia. Pneumonia is a common presentation in melioidosis caused by B. pseudomallei. In such patients with pneumonia, a bout of massive hemoptysis can be fatal. Culture is the gold standard diagnostic method in melioidosis. Burholderia pseudomallei can be easily differentiated from other organisms like Pseudomonas spp. and B. cepacia by Gram stain, methylene blue stain, and screening for polymyxin sensitivity. Early diagnosis and appropriate antibiotic therapy are warranted in melioidosis. AcknowledgmentWe acknowledge Professor and Head of department of Internal Medicine, Dr. Paranthaman (MD), Government Kilpauk Medical College, Chennai, for his guidance and support and providing the necessary details of the patient in publishing this article Conflict of interestThe authors declare that there is no conflict of interest. ReferencesBenoit, T.J., Blaney, D.D., Doker, T.J., Gee, J.E., Elrod, M.G., Rolim, D.B., Inglis, T.J., Hoffmaster, A.R., Bower, W.A. and Walke, H.T. 2015. A review of melioidosis cases in the Americas. Ame. J. Trop. Med. Hyg. 93(6), 1134. Currie, B.J. 2015. Melioidosis: evolving concepts in epidemiology, pathogenesis, and treatment. Semin. Respir. Crit. Care. Med. 36(01), 111–125. Doker, T.J., Quinn, C.L., Salehi, E.D., Sherwood, J.J., Benoit, T.J., Elrod, M.G., Gee, J.E., Shadomy, S.V., Bower, W.A., Hoffmaster, A.R. and Walke, H.T. 2014. Case report: fatal Burkholderia pseudomallei infection initially reported as a Bacillus species, Ohio, 2013. Am. J. Trop. Med. Hyg. 91(4), 743. Fertitta, L., Monsel, G., Torresi, J. and Caumes, E. 2019. Cutaneous melioidosis: a review of the literature. Int. J. Dermatol. 58(2), 221–227. Foong, Y.C., Tan, M. and Bradbury, R.S. 2014. Melioidosis: a review. Rural. Remote. Health. 14(4), 114–129. Hodgson, K., Engler, C., Govan, B., Katheen, N. and Norton, R. 2009. Comparison of routine bench and molecular diagnostic methods in identification of Burkholderia pseudomallei. J. Clin. Microbiol. 47(5), 1578–1580. Klietmann, W.F. and Ruoff, K.L. 2001. Bioterrorism: implications for the clinical microbiologist. Clin. Microbiol. Rev. 14(2), 364–381. Lau, S.K., Sridhar, S., Ho, C.C., Chow, W.N., Lee, K.C., Lam, C.W., Yuen, K.Y. and Woo, P.C. 2015. Laboratory diagnosis of melioidosis: past, present and future. Exp. Biol. Med. 240(6), 742–751. Limmathurotsakul, D., Jamsen, K., Arayawichanont, A., Simpson, J.A., White, L.J., Lee, S.J., Wuthiekanun, V., Chantratita, N., Cheng, A., Day, N.P. and Verzilli, C. 2010. Defining the true sensitivity of culture for the diagnosis of melioidosis using Bayesian latent class models. PloS One 5(8), e12485. Lipsitz, R., Garges, S., Aurigemma, R., Baccam, P., Blaney, D.D., Cheng, A.C., Currie, B.J., Dance, D., Gee, J.E., Larsen, J. and Limmathurotsakul, D. 2012. Workshop on treatment of and postexposure prophylaxis for Burkholderia pseudomallei and B. mallei infection, 2010. Emerg. Infect. Dis. 18(12), e2. Suttisunhakul, V., Wuthiekanun, V., Brett, P.J., Khusmith, S., Day, N.P., Burtnick, M.N., Limmathurotsakul, D. and Chantratita, N. 2016. Development of rapid enzyme-linked immunosorbent assays for detection of antibodies to Burkholderia pseudomallei. J. Clin. Microbiol. 54(5), 1259–1268. Trinh, T.T., Hoang, T.S., Tran, D.A., Trinh, V.T., Nguyen, T.T., Hoang, S.N., Krumkamp, R., Nguyen, L.T., May, J., Doan, P.M., Do, C.D., Que, T.A. and Steinmetz, I. 2018. A simple laboratory algorithm for diagnosis of melioidosis in resource-constrained areas: a study from north-central Vietnam. Clin. Microbiol. Infect. 24(1), 84. Vidyalakshmi, K., Chakrapani, M., Shrikala, B., Damodar, S., Lipika, S. and Vishal, S. 2008. Tuberculosis mimicked by melioidosis. Int. J. Tubercu. Lung. Dis. 12(10), 1209–1215. Whitmore A. An account of a glanders-like disease occurring in Rangoon. Epidemiology & Infection. 1913 Apr;13(1):1-34. Wiersinga, W.J., Virk, H.S., Torres, A.G., Currie, B.J., Peacock, S.J., Dance, D.A. and Limmathurotsakul, D. 2018. Melioidosis. Nat. Rev. Dis. Prim. 4(1), 1–22. Wiersinga, W.J., Virk, H.S., Torres, A.G., Currie, B.J., Peacock, S.J., Dance, D.A.B. and Limmathurotsakul, D. 2018. Melioidosis. Nat. Rev. Dis. Prim. 4(17107), 17107. | ||
| How to Cite this Article |
| Pubmed Style Dhason TM, Ravinder T, Krishnan U, Ammanarasimman P, Kamalasekaran LT, Chellaiah A. A case of melioidosis with hemoptysis: Strategies to isolate and identify Burkholderia pseudomallei. J Microbiol Infect Dis. 2023; 13(2): 87-89. doi:10.5455/JMID.2023.v13.i2.6 Web Style Dhason TM, Ravinder T, Krishnan U, Ammanarasimman P, Kamalasekaran LT, Chellaiah A. A case of melioidosis with hemoptysis: Strategies to isolate and identify Burkholderia pseudomallei. https://www.jmidonline.org/?mno=302657426 [Access: July 03, 2025]. doi:10.5455/JMID.2023.v13.i2.6 AMA (American Medical Association) Style Dhason TM, Ravinder T, Krishnan U, Ammanarasimman P, Kamalasekaran LT, Chellaiah A. A case of melioidosis with hemoptysis: Strategies to isolate and identify Burkholderia pseudomallei. J Microbiol Infect Dis. 2023; 13(2): 87-89. doi:10.5455/JMID.2023.v13.i2.6 Vancouver/ICMJE Style Dhason TM, Ravinder T, Krishnan U, Ammanarasimman P, Kamalasekaran LT, Chellaiah A. A case of melioidosis with hemoptysis: Strategies to isolate and identify Burkholderia pseudomallei. J Microbiol Infect Dis. (2023), [cited July 03, 2025]; 13(2): 87-89. doi:10.5455/JMID.2023.v13.i2.6 Harvard Style Dhason, T. M., Ravinder, . T., Krishnan, . U., Ammanarasimman, . P., Kamalasekaran, . L. T. & Chellaiah, . A. (2023) A case of melioidosis with hemoptysis: Strategies to isolate and identify Burkholderia pseudomallei. J Microbiol Infect Dis, 13 (2), 87-89. doi:10.5455/JMID.2023.v13.i2.6 Turabian Style Dhason, Therese Mary, Thyagarajan Ravinder, Usha Krishnan, Pavithra Ammanarasimman, Lavanya T. Kamalasekaran, and Amutha Chellaiah. 2023. A case of melioidosis with hemoptysis: Strategies to isolate and identify Burkholderia pseudomallei. Journal of Microbiology and Infectious Diseases, 13 (2), 87-89. doi:10.5455/JMID.2023.v13.i2.6 Chicago Style Dhason, Therese Mary, Thyagarajan Ravinder, Usha Krishnan, Pavithra Ammanarasimman, Lavanya T. Kamalasekaran, and Amutha Chellaiah. "A case of melioidosis with hemoptysis: Strategies to isolate and identify Burkholderia pseudomallei." Journal of Microbiology and Infectious Diseases 13 (2023), 87-89. doi:10.5455/JMID.2023.v13.i2.6 MLA (The Modern Language Association) Style Dhason, Therese Mary, Thyagarajan Ravinder, Usha Krishnan, Pavithra Ammanarasimman, Lavanya T. Kamalasekaran, and Amutha Chellaiah. "A case of melioidosis with hemoptysis: Strategies to isolate and identify Burkholderia pseudomallei." Journal of Microbiology and Infectious Diseases 13.2 (2023), 87-89. Print. doi:10.5455/JMID.2023.v13.i2.6 APA (American Psychological Association) Style Dhason, T. M., Ravinder, . T., Krishnan, . U., Ammanarasimman, . P., Kamalasekaran, . L. T. & Chellaiah, . A. (2023) A case of melioidosis with hemoptysis: Strategies to isolate and identify Burkholderia pseudomallei. Journal of Microbiology and Infectious Diseases, 13 (2), 87-89. doi:10.5455/JMID.2023.v13.i2.6 |