| Research Article | ||
J Microbiol Infect Dis. 2023; 13(3): 139-147 J. Microbiol. Infect. Dis., (2023), Vol. 13(3): 139–147 Original Research Influencing factors of central venous catheter-associated bloodstream infection in patients with emergency hemorrhagic shockJianping Zhu, Yanxin Xu, Shaohong Wu, Feiyao Wang, Weixing Zhang and Ruilan Wang*Department of Critical Care Medicine, Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China *Corresponding Author: Ruilan Wang. Department of Critical Care Medicine, Shanghai General Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China. Email: wangyusun [at] hotmail.com Submitted: 05/05/2023 Accepted: 05/08/2023 Published: 30/09/2023 © 2023 Journal of Microbiology and Infectious Diseases
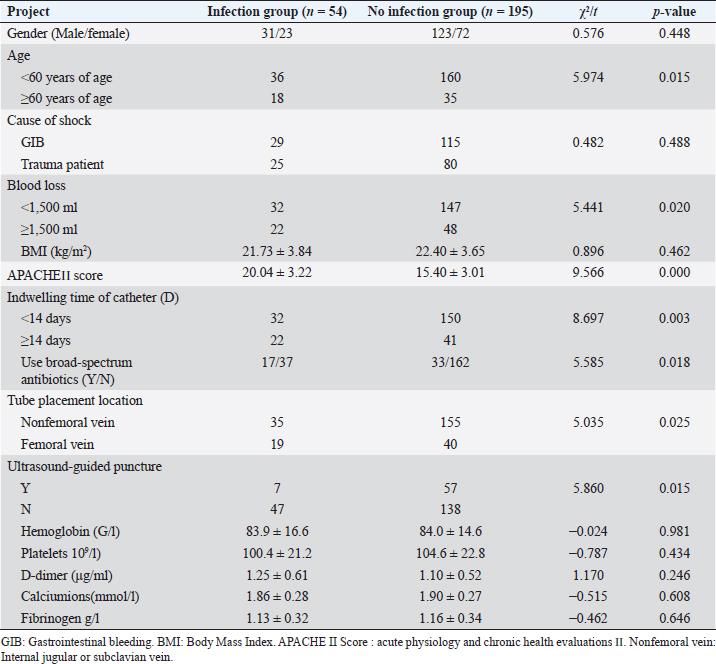
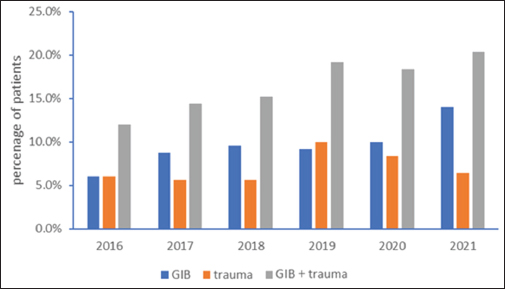
AbstractObjective: To investigate and analyze the influencing factors of central venous catheter-associated bloodstream infection in patients with emergency hemorrhagic shock. Methods: According to the inclusion criteria, 249 patients with hemorrhagic shock and central venous catheterization admitted to the emergency department of Shanghai First People’s Hospital from June 2016 to 2022 were retrospectively analyzed, and divided into the infection group (54 cases), and the noninfection group (195 cases) according to whether catheter-related bloodstream infection occurred. Multivariate logistic regression was used to analyze the relationship between central venous catheter-related bloodstream infection and various influencing factors. The etiological and drug susceptibility test data of peripheral venous blood culture, puncture site samples, and catheter tip samples from patients with central venous catheter-associated bloodstream infection were statistically analyzed. Results: There were significant differences in age, catheterization location, ultrasound-guided puncture, acute physiology and chronic health evaluations II (APACHE II) score, catheter indwelling time, and application of broad-spectrum antibiotics between the infected group and the noninfected group (p < 0.05). Staphylococcus epidermidis accounted for 44.4%, Escherichia coli 24.1%, and fungi 11.1% in 54 infected patients. The lowest resistance rate of Gram-positive cocci was to vancomycin (3.4%), followed by tetracycline 34.5%, and the highest resistance rate was to amoxicillin 100.0%. Conclusion: The risk factors of central venous catheter-related bloodstream infection in patients with emergency hemorrhagic shock include advanced age, femoral vein catheterization, nonultrasound-guided puncture, high APACHE II score, catheter retention time, and application of broad-spectrum antibiotics. Staphylococcus epidermidis the common pathogen of central venous catheterization-related infection in emergency hemorrhagic shock patients. Keywords: Hemorrhagic shock, Central vein catheterization, Catheter-related bloodstream infection, Etiology, Drug resistance. IntroductionIn recent years, the incidence of catheter-related blood stream infection (CRBSI) has declined, but remains high, with the highest morbidity and mortality, especially in the intensive care unit (Badia-Cebada et al., 2022; Verberk et al., 2023). CRBSI patients lack characteristic clinical manifestations, usually, fever is the primary manifestation, and redness, swelling, pain, and pus at the insertion site are highly specific. Other clinical manifestations include hemodynamic instability, catheter dysfunction, and sudden sepsis (Tripathi et al., 2021). The occurrence of catheter-related bloodstream infection in patients not only prolongs hospital stay, and increases medical costs, but also increases the difficulty of clinical treatment, thus adversely affecting the prognosis of patients (Forrester et al., 2022; Nelson et al., 2022). Therefore, in recent years, how to reduce the infection rate of CRBSI is a topic that clinicians are actively studying (Rickard et al., 2021; Eturajulu et al., 2022; Kotwal et al., 2022; Lin et al., 2022). However, there are few reports on the occurrence of CRBSI in patients with hemorrhagic shock in an emergency. Patients with hemorrhagic shock are in urgent condition, so there is not enough time to make a comprehensive assessment of their condition. The emergency physicians are busy with rescuing patients and neglect strict aseptic operation standards. In addition, the occurrence of catheter-related infection and the distribution of pathogenic bacteria in patients with hemorrhagic shock in the emergency department also deserves our attention. In this study, 249 patients diagnosed with emergency hemorrhagic shock and receiving deep vein catheterization in our hospital from June 2016 to 2022 were selected as the research objects, mainly patients with hemorrhagic shock caused by acute gastrointestinal bleeding and trauma, and the influencing factors and pathogenic bacteria were analyzed, aiming to clarify the risk factors and pathogen distribution of catheter-related infection in patients with emergency hemorrhagic shock central venous catheterization and to provide sufficient and reliable evidence for their treatment. Materials and MethodsSubjectsA total of 249 patients with hemorrhagic shock treated in our hospital from June 2016 to 2022 were selected, all of whom received central venous catheterization in the emergency room of our hospital and met the inclusion criteria. Criteria for diagnosis of hemorrhagic shock: (1) systolic blood pressure <90 mmHg or more than 30% lower than the basal level or pulse pressure <30 mmHg; (2) conscious disorders, such as irritability, indifferent expression or vague consciousness, and coma; (3) cold and wet limbs or mottled skin, pale mucous membranes or cyanosis; and (4) Pulse >100 beats/minute or not palpable. When the first is combined with any of the last three, hemorrhagic shock is diagnosed. Inclusion criteria: (1) patients who received central venous catheterization in the emergency department of our hospital and met the diagnostic criteria for hemorrhagic shock; (2) age ≥20 years old; (3) central venous catheter was left in place for more than 48 hours; (4) when the same patient has multiple diagnoses, only the first diagnosis is selected; and (5) patients with complete clinical records. Exclusion criteria: (1) patients with organic lesions of heart, lung, liver, kidney, and other organs; (2) patients who received immunosuppressive drugs or antibacterial drugs within 1 month before deep vein catheterization; (3) patients who died, were transferred to another hospital or gave up treatment during hospitalization; and (4) patients with infection symptoms such as fever or chills 48 hours before deep vein catheterization. This study complies with the Declaration of Helsinki. Collection of medical history dataThe clinical medical records of 249 patients were retrospectively analyzed, and the cases were retrieved by the medical record inquiry system of the hospital. Two researchers entered the forms and one reviewed them. The specific content of relevant data included gender, age, location of catheterization, blood loss, cause of shock, body mass index (BMI), acute physiology and chronic health evaluations II (APACHE II) score, time of indentation of deep vein catheter, application of broad-spectrum antibiotics, ultrasound-guided puncture or not, and related blood test results. According to the occurrence of catheter-related bloodstream infection, 54 cases were in the infection group and 195 cases were in the noninfection group. Specimen collection and pathogen culture methodsAll catheters were subjected to routine bacterial culture and peripheral blood culture was retained at the time of removal. After disinfecting the skin around the CVC puncture site, pull out the catheter and cut the proximal tip 4–5 cm with sterile scissors. Samples were collected and placed in a sterile container and sent to the microbiological laboratory within 15 minutes at room temperature. The tip of the catheter was cultured by semi-quantitative culture technique. The BactecTMFX40 blood culture instrument produced by Becton Dickinson Company in the United States was used for blood culture, and the pathogen was identified by the phoenix 100 automatic bacterial identification drug sensitive system produced by Becton Dickinson Company in the United States. Diagnostic criteria for CRBSI (Mermel et al., 2009). According to the Infectious Disease Society of America guidelines, a patient who presents with symptoms of infection and rules out infection at other sites has a semi-quantitative population count >15 cfu or a quantitative population count >102 cfu at the catheter tip, and meets one of the following conditions: (1) the pathogens cultured at the tip of the catheter were at least the same as the results of one peripheral blood culture; (2) blood was collected from two different catheter cavities for culture, and the colony count ratio of the two cavities was >3:1; (3) the ratio of colony count between blood culture with catheter and peripheral venous blood culture was >3:1; and (4) the time of positive results of catheter blood culture should be at least 2 hours earlier than that of peripheral venous blood culture. Statistical analysisSPSS 21.0 software was used to analyze the data. The measurement data is represented by mean ± SD (`χ ± s), and the counting data was expressed as a percentage. The chi-square test was used for comparison between groups, and Logistic regression was used for multivariate analysis. For p < 0.05, the difference was considered statistically significant. Ethical approval and consent to participateThis study complies with the Declaration of Helsinki. ResultsFrom June 2016 to 2022, a total of 249 emergency patients with hemorrhagic shock who met the inclusion criteria were admitted to our hospital, mainly including patients with trauma and gastrointestinal bleeding. The number of emergency patients with hemorrhagic shock was increasing year by year, especially the bleeding shock caused by gastrointestinal bleeding (Fig. 1). Baseline data for both groups: There was no significant difference between the infected group and the noninfected group in gender, cause of shock, BMI and related blood test indexes (p > 0.05), while there were statistically significant age differences, location of central venous catheters, blood loss, APACHE II score, the duration of deep venous catheter indwelling,application of broad-spectrum antibiotics and ultrasound-guided puncture between the two groups (p < 0.05) (Table 1). Table 1. Univariate analysis of catheter-related infection in patients with emergency hemorrhagic shock with deep vein catheterization.
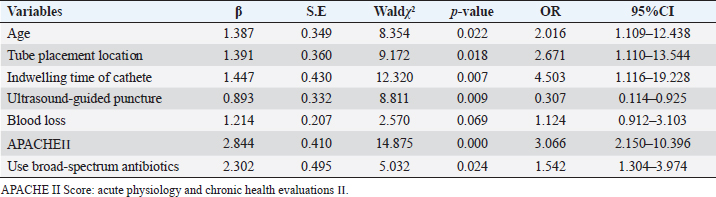
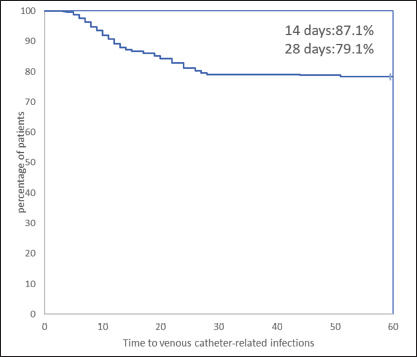
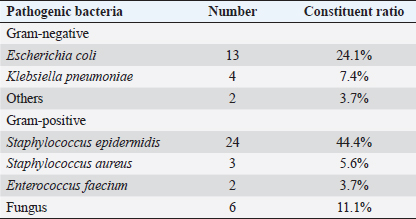
Multivariate logistic regression analysis of risk factors of central venous catheter-associated bloodstream infection: central venous catheter-associated bloodstream infection was taken as the dependent variable, and assigned: infection=1, no infection=0. Age, location of central vein catheterization, blood loss, APACHE II score, indwelling time of deep vein catheter, application of broad-spectrum antibiotics, and ultrasound-guided puncture were independent variables. Age, APACHE II score at admission, and duration of the indwelling of the catheter were input as original values and other independent variables were input as yes=1, no=0. Age, location of central venous catheterization, APACHE II score, indwelling time of deep venous catheterization, application of broad-spectrum antibiotics, and ultrasound-guided puncture were the risk factors for central venous catheter-related bloodstream infection in patients with hemorrhagic shock (p < 0.05) (Table 2). There were 54 cases of CRBSI with an infection rate of 21.7%. The occurrence time of CRBSI is shown in Figure 2. The noncatheter-associated infection rate was 87.1% at 14 days and 79.1% at 28 days after central vein implantation. Distribution of pathogenic bacteria: the most pathogenic bacteria were Staphylococcus epidermidis (44.4%), Escherichia coli (24.1%), and fungi (11.1%), as shown in Table 3. Table 2. Logistic analysis of multiple factors affecting the occurrence of catheter-related infection in patients with hemorrhagic shock with deep vein catheterization.
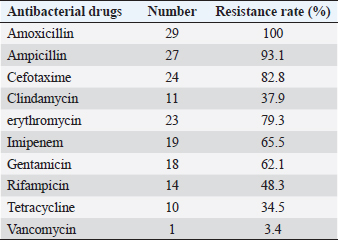
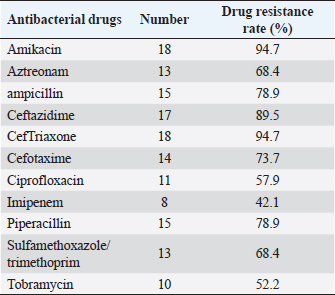
Fig. 1. Occurrence of patients with emergency 335 shock in our hospital in the past 6 years. Drug sensitivity test: the lowest resistance rate of Gram-positive cocci was vancomycin (3.4%), followed by tetracycline (34.5%). The highest resistance rate was to amoxicillin at 100.0%. The drug resistance rate of Gram-negative bacilli to amikacin and ceftriaxone was very high (94.7%), as shown in Tables 4 and 5. Therapeutic effect: according to the etiological detection and drug sensitivity results of the patients, the patients were treated with appropriate antibacterial drugs for (8.1 ± 1.3) days. The infection of all patients was controlled, clinical symptoms disappeared, and the bacterial culture results were negative. Finally, the patients were cured and discharged from the hospital. DiscussionHemorrhagic shock patients are in critical condition with high mortality (Faria et al., 2022; Gauss et al., 2022); rapid diagnosis and treatment is the key, the first need to receive a large number of rapid blood transfusion, fluid replacement, and central venous catheterization is one of the essential operations, with good catheter elasticity, long retention time and simple operation, with the help of ultrasound guidance, can significantly improve the success rate of puncture and reduce related injuries, has been widely used in the clinical rescue of patients with shock, heart failure, dehydration and insufficient blood volume (Czarnik et al., 2023). However, as an invasive technique, deep vein catheterization has a high incidence of catheter-related infection (Smit et al., 2022). The enrolled population in this study were patients with hemorrhagic shock who came to our hospital, mainly including patients with gastrointestinal bleeding and traumatic shock, and their condition was urgent. As can be seen from Figure 1, the number of patients with hemorrhagic shock coming to our hospital has increased year by year in the past 6 years, especially for patients with gastrointestinal bleeding, although this only reflects the occurrence of hemorrhagic shock in our hospital. In this study, 54 cases of CRBSI were diagnosed, and the infection rate was about 21.7%, which was significantly higher than the 13% reported by Bisanti et al. (2023) (CRBSI in ICU). This indicates a higher incidence of CRBS in patients with hemorrhagic shock in the emergency department. The analysis of basic data for infected and noninfected groups (Table 1) and the results of Logistic regression analysis (Table 2) showed that age, location of central vein catheterization, APACHE II score, duration of deep vein catheterization, application of broad-spectrum antibiotics and ultrasound-guided puncture were independent risk factors for CRBSI. The decrease of the body’s immunity caused by the increase in age and higher APACHE II score may be the important reasons for the occurrence of intravenous catheterization infection in patients (Rickard et al., 2021). With the increase of catheter indwelling time, serum proteins, fibrin, and other substances in the blood are easily deposited on the surface of the catheter. The above-mentioned sediments are ideal culture medium for pathogenic bacteria, playing a role of protecting pathogenic bacteria from being killed by immune cells and antibacterial drugs, thus promoting the release of a large number of pathogenic bacteria into the blood, and finally causing infection. In the multivariate Logistic regression analysis of this study, the OR value of catheter indwelling time was 4.503. It can be seen from Figure 2 that the noncatheter related infection rate was 87.1% on the 14th day and 79.1% on the 28th day after implantation of central venous catheter, indicating that the longer the indwelling time of catheter, the greater the possibility of catheter-related bloodstream infection. Polderman and Girbes (2002) and Pitiriga et al.’s (2022) study also showed that catheter indwelling time was an independent risk factor for catheter-related infection, so the central venous catheter should be removed as soon as possible. This study suggested that the use of antibiotics was also an important risk factor for CLABSI in patients with hemorrhagic shock (OR=1.542), and the use of broad-spectrum antibiotics could easily lead to the generation of drug-resistant strains, thereby increasing the probability of infection. Rinke et al. (2021) showed that a patient’s history of antibacterial therapy was identified as a risk factor for catheter-associated bloodstream infections. Some patients with bacterial colonization may end up infected due to unreasonable timing of administration, drug selection, and medication duration. Guidelines for CLABSI prevention and control issued by the CENTERS for disease control and prevention and the Advisory Committee on Hospital Infection Control Practices in June 2017, also explicitly stated that routine systemic prophylactic use of antibiotics should not be used to prevent catheter bacterial colonization before or during endovascular catheterization (Mermel et al., 2009). Regarding the role of ultrasound in central venous catheterization, it has been reported that ultrasound-guided central venous catheterization can reduce the incidence of catheter-related bloodstream infections (Timsit et al., 2020; Adrian et al., 2022). However, Khera et al. (2023) suggested that ultrasound-guided central vein catheterization was not associated with catheter-associated bloodstream infection. Buetti et al. (2021) believes that improper ultrasound procedures can increase the incidence of catheter-associated infections. In this study, it was found that the rate of catheter infection in ultrasound-guided central venous catheterization was lower than that in nonultrasound-guided catheterization. We believe that ultrasound guidance can reduce the incidence of catheter bloodstream infection as long as the strict aseptic operation is performed, which may be mainly due to the higher success rate of puncture under ultrasound, which reduces the number of puncture and tissue damage, thus reducing the probability of catheter infection. In this study, it was also found that the incidence of infection varied with the location of central vein catheterization. The incidence of infection of the internal jugular vein or subclavian vein catheterization was lower than that of femoral vein catheterization, which was similar to the findings of Bisanti et al. (2023) and Heidenreich et al. (2022). Parienti et al.’s (2015) study identified a low subclavian vein access infection rate but a high likelihood of pneumothorax. Therefore, jugular vein access can be used as the preferred site for catheterization in the absence of obvious contraindications.
Fig. 2. The time of catheter-related infection after central venous catheter in patients with hemorrhagic shock. The rate of noncatheter-related infection was 87.1% at 14 days and 79.1% at 28 days after central venous implantation. Table 3. Distribution and constituent ratio of pathogens infected by patients (%).
Table 4. Drug resistance rate of 29 strains of Gram-positive cocci to antibiotics (%).
Table 5. Drug resistance rate of 19 strains of Gram-negative bacilli to antibiotics (%).
In this study, pathogenic strains were cultured in all 54 patients with catheter-associated infection samples, and the main pathogenic bacteria were Gram-positive bacteria (53.7%), which was related to relevant studies the results are similar (Ripa et al., 2018; Calò et al., 2020; San-Juan et al., 2022). In addition, drug sensitivity results showed that Gram-positive cocci were the most sensitive to vancomycin, San-Juan et al. (2016) believes that vancomycin should continue to be the primary treatment option for Gram-positive catheter-associated bloodstream infections. In this study, amoxicillin was found to have the highest resistance rate, followed by ampicillin. Therefore, in clinical practice, antibiotics are selected according to patients’ drug sensitivity. Gram-negative bacilli had the highest sensitivity to imipenem, followed by tobramycin, but the drug resistance rate to amikacin and ceftriaxone was relatively high, which has a certain reference value for guiding clinical empirical drug use. Current guidelines recommend antibiotic treatment for at least 7–14 days, and recent data suggest that appropriate antibiotic treatment for 7 days or less may be as effective as a longer course of treatment for catheter infection after removal of the central venous catheter (Muff et al., 2021; Ruiz-Ruigómez and Aguado, 2021). In this study, antibiotics were used for (8.1 ± 1.3) days, infection of all patients was controlled, clinical symptoms disappeared, and peripheral blood bacterial culture results were negative. Zhong et al. (2022) believes that early replacement of central venous catheters is safe in patients with CRBSI. There should be no delay in replacing a Central Line in patients who still need it. This study was a retrospective analysis, which had certain limitations, such as whether to disinfect hands before puncture, the number of punctures, and the working years of surgeons, which could not be counted. This is a single-center study with a relatively small number of cases, and the results may be biased. In the future, we will further expand the sample size and conduct prospective studies to provide more sufficient and reliable evidence for the prevention of CRBSI in patients with hemorrhagic shock. In conclusion, the incidence of catheter-related infection in patients with deep vein catheterization in hemorrhagic shock is still high, and Gram-positive bacteria is the main pathogen, which is related to the patient’s age, catheterization location, catheter indentation time, APACHE II score, application of broad-spectrum antibiotics, and ultrasound-guided puncture. To reduce the incidence of catheter-related infection in patients with hemorrhagic shock deep vein catheterization, intervention measures should be formulated according to the risk factors of central venous catheter-related bloodstream infection in patients with hemorrhagic shock. In clinical work, physicians should strengthen the evaluation of the condition of patients with hemorrhagic shock, select the internal jugular vein or subclavian vein for puncture as far as possible, conduct puncture under the guidance of ultrasound as far as possible, remove the deep vein catheter as soon as possible, use less or no broad-spectrum antibiotics, and use antibiotics reasonably according to the results of bacterial blood culture when applying antibiotics. Author contributionsJianping Zhu: This author conceived and designed the study, wrote the manuscript, and has approved the publication of this version. He is accountable for the accuracy and integrity of the work. Yanxin Xu: This author participated in data collection, performed the data analysis, wrote the manuscript, and has approved the publication of this version. He is accountable for the accuracy and integrity of the work. Shaohong Wu: This author participated in data collection and performed the data analysis. Feiyao Wang: This author participated in the study design, data interpretation, and manuscript editing. Weixing Zhang: This author participated in the study design, data interpretation, and manuscript editing. Ruilan Wang: This author conceived and designed the study, wrote the manuscript, and approved the publication of this version. All authors read and approved the final manuscript. FundingThis research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. Conflict of interestThe authors declare that they have no competing interests. ReferencesAdrian, M., Borgquist, O., Kröger, T., Linné, E., Bentzer, P., Spångfors, M., Åkeson, J., Holmström, A., Linnér, R. and Kander, T. 2022. Mechanical complications after central venous catheterisation in the ultrasound-guided era: a prospective multicentre cohort study. Br. J. Anaesth. 129(6), 843–850. Badia-Cebada, L., Peñafiel, J., Saliba, P., Andrés, M., Càmara, J., Domenech, D., Jiménez-Martínez, E., Marrón, A., Moreno, E., Pomar, V. and Vaqué, M. 2022. Trends in the epidemiology of catheter-related bloodstream infections;towards a paradigm shift, Spain, 2007 to 2019. Euro. Surveill. 27(19), 2100610. Bisanti, A., Giammatteo, V., Bello, G., Giannarelli, D., Montini, L., Tanzarella, E.S., Carelli, S., Bongiovanni, F., D’Inzeo, T., Fiori, B. and Grieco, D.L. 2023. Usefulness of differential time to positivity between catheter and peripheral blood cultures for diagnosing catheter-related bloodstream infection: data analysis from routine clinical practice in the intensive care unit. J. Crit. Care. 75, 154259. Buetti, N., Mimoz, O., Mermel, L., Ruckly, S., Mongardon, N., Dupuis, C., Mira, J.P., Lucet, J.C., Mégarbane, B., Bailly, S. and Parienti, J.J. 2021. Ultrasound guidance and risk for central venous catheter-related infections in the intensive care unit: a post hoc analysis of individual data of 3 multicenter randomized trials. Clin. Infect. Dis. 73(5), e1054–e1061. Calò, F., Retamar, P., Martínez Pérez-Crespo, P.M., Lanz-García, J., Sousa, A., Goikoetxea, J., Reguera-Iglesias, J.M., León, E., Armiñanzas, C., Mantecón, M.A. and Rodríguez-Baño, J. 2020. Catheter-related bloodstream infections:predictive factors for Gram-negative bacteria aetiology and 30 day mortality in a multicentre prospective cohort. J. Antimicrob. Chemother. 75(10), 3056–3061. Czarnik, T., Czuczwar, M., Borys, M., Chrzan, O., Filipiak, K., Maj, M., Marszalski, M., Miodonska, M., Molsa, M., Pietka, M. and Piwoda, M. 2023. Ultrasound-guided infraclavicular axillary vein versus internal jugular vein cannulation in critically Ill mechanically ventilated patients: a randomized trial. Crit. Care. Med. 51(2), e37–e44. Eturajulu, R.C., Ng, K.H., Tan, M.P., Zakaria, M.I., Chinna, K., Ponnampalavanar, S., Vijayananthan, A., Yaakup, N.A. and Chung, E. 2022. Quality improvement report: safety program for prevention of central line-associated bloodstream infections. Radiographics 42(7), E216–E223. Faria, I., Thivalapill, N., Makin, J., Puyana, J.C. and Raykar, N. 2022. Bleeding, hemorrhagic shock, and the global blood supply. Crit. Care. Clin. 38(4), 775–793. Forrester, J.D., Maggio, P.M. and Tennakoon, L. 2022. Cost of health care-associated infections in the United States. J. Patient. Saf. 18(2), e477–e479. Gauss, T., Richards, J.E., Tortù, C., Ageron, F.X., Hamada, S., Josse, J., Husson, F., Harrois, A., Scalea, T.M., Vivant, V. and Meaudre, E. 2022. Association of early norepinephrine administration with 24-hour mortality among patients with blunt trauma and hemorrhagic shock. JAMA. Netw. Open. 5(10), e2234258. Heidenreich, D., Hansen, E., Kreil, S., Nolte, F., Jawhar, M., Hecht, A., Hofmann, W.K. and Klein, S.A. 2022. The insertion site is the main risk factor for central venous catheter-related complications in patients with hematologic malignancies. Am. J. Hematol. 97(3), 303–310. Khera, S., Kumar, A., Parikh, B., Simalti, A.K., Davera, S., Mahajan, P. and Dhingra, S. 2023. Safety and outcome of ultrasound-guided tunneled central venous catheter in children with cancers from low middle-income country: a prospective study. Pediatr. Blood. Cancer. 70(1), e30029. Kotwal, S., Cass, A., Coggan, S., Gray, N.A., Jan, S., McDonald, S., Polkinghorne, K.R., Rogers, K., Talaulikar, G., Di Tanna, G.L. and Gallagher, M. 2022. Multifaceted intervention to reduce haemodialysis catheter related bloodstream infections: REDUCCTION stepped wedge, cluster randomised trial. BMJ 377, e069634. Lin, M.R., Chang, P.J., Hsu, P.C., Lin, C.S., Chiu, C.H. and Chen, C.J. 2022. Comparison of efficacy of 2% chlorhexidine gluconate-alcohol and 10% povidone-iodine-alcohol against catheter-related bloodstream infections and bacterial colonization at central venous catheter insertion sites: a prospective, single-center, open-label, crossover study. J. Clin. Med. 11(8), 2242. Mermel, L.A., Allon, M., Bouza, E., Craven, D.E., Flynn, P., O’Grady, N.P., Raad, I.I., Rijnders, B.J., Sherertz, R.J. and Warren, D.K. 2009. Clinical practice guidelines for the diagnosis and management of intravascular catheter-related infection:2009 update by the Infectious Diseases Society of America. Clin. Infect. Dis. 49(1), 1–45. Muff, S., Tabah, A., Que, Y.A., Timsit, J.F., Mermel, L., Harbarth, S. and Buetti, N. 2021. Short-course versus long-course systemic antibiotic treatment for uncomplicated intravascular catheter-related bloodstream infections due to Gram-negative bacteria, enterococci or coagulase-negative staphylococci: a systematic review. Infect. Dis. Ther. 10(3), 1591–1605. Nelson, R.E., Hyun, D., Jezek, A. and Samore, M.H. 2022. Mortality, length of stay, and healthcare costs associated with multidrug-resistant bacterial infections among elderly hospitalized patients in the United States. Clin. Infect. Dis. 74(6), 1070–1080. Parienti, J.J., Mongardon, N., Mégarbane, B., Mira, J.P., Kalfon, P., Gros, A., Marqué, S., Thuong, M., Pottier, V., Ramakers, M. and Savary, B. 2015. Intravascular complications of central venous catheterization by insertion site. N. Engl. J. Med. 373(13), 1220–1229. Pitiriga, V., Bakalis, J., Kampos, E., Kanellopoulos, P., Saroglou, G. and Tsakris, A. 2022. Duration of central venous catheter placement and central line-associated bloodstream infections after the adoption of prevention bundles:a two-year retrospective study. Antimicrob. Resist. Infect. Control. 11(1), 96. Polderman, K.H. and Girbes, A.R. 2002. Central venous catheter use. Part 2: infectious complications. Intensive. Care. Med. 28(1), 18–28. Rickard, C.M., Marsh, N.M., Larsen, E.N., McGrail, M.R., Graves, N., Runnegar, N., Webster, J., Corley, A., McMillan, D., Gowardman, J.R. and Long, D.A. 2021. Effect of infusion set replacement intervals on catheter-related bloodstream infections (RSVP):a randomised, controlled, equivalence (central venous access device)-noninferiority (peripheral arterial catheter) trial. Lancet 397(10283), 1447–1458. Rinke, M.L., Heo, M., Saiman, L., Bundy, D.G., Rosenberg, R.E., DeLaMora, P., Rabin, B., Zachariah, P., Mirhaji, P., Ford, W.J. and Obaro-Best, O. 2021. Pediatric ambulatory central line-associated bloodstream infections. Pediatrics 147(1), e20200524. Ripa, M., Morata, L., Rodríguez-Núñez, O., Cardozo, C., Puerta-Alcalde, P., Hernández-Meneses, M., Ambrosioni, J., Linares, L., Bodro, M., Valcárcel, A. and Casals, C. 2018. Short-term peripheral venous catheter-related bloodstream infections: evidence for increasing prevalence of Gram-negative microorganisms from a 25-year prospective observational study. Antimicrob. Agents. Chemother. 62(11), e00892–e00818. Ruiz-Ruigómez, M. and Aguado JM. 2021. Duration of antibiotic therapy in central venous catheter-related bloodstream infection due to Gram-negative bacilli. Curr. Opin. Infect. Dis. 34(6), 681–685. San-Juan, R., Ruiz-Ruigómez, M. and Aguado, J.M. 2022. How to manage central venous catheter-related bloodstream infections due to Gram-negative bacilli? Curr. Opin. Infect. Dis. 35(6), 583–588. San-Juan, R., Viedma, E., Chaves, F., Lalueza, A., Fortún, J., Loza, E., Pujol, M., Ardanuy, C., Morales, I., de Cueto, M. and Resino-Foz, E. 2016. High MICs for vancomycin and daptomycin and complicated catheter-related bloodstream infections with methicillin-sensitive Staphylococcus aureus. Emerg. Infect. Dis. 22(6), 1057–1066. Smit, J.M., Exterkate, L., Van Tienhoven, A.J., Haaksma, M.E., Heldeweg, M.L., Fleuren, L., Thoral, P., Dam, T.A., Heunks, L., Gommers, D. and Cremer, O.L. 2022. Incidence, risk factors,and outcome of suspected central venous catheter-related infections in critically ill COVID-19 patients:a multicenter retrospective cohort study. Shock 58(5), 358–365. Timsit, J.F., Baleine, J., Bernard, L., Calvino-Gunther, S., Darmon, M., Dellamonica, J., Desruennes, E., Leone, M., Lepape, A., Leroy, O. and Lucet, J.C. 2020. Expert consensus-based clinical practice guidelines management of intravascular catheters in the intensive care unit. Ann. Intensive. Care. 10(1), 118. Tripathi S, Kumar S, Kaushik S. The practice and complications of midline catheters: a systematic review. Crit. Care. Med. 49(2), e140–e150. Verberk, J.D., van der Kooi, T.I., Kampstra, N.A., Reimes, N., van Rooden, S.M., Hopmans, T.E., Geerlings, S.E. and de Greeff, S.C. 2023. Healthcare-associated infections in Dutch hospitals during the COVID-19 pandemic. Antimicrob. Resist. Infect. Control. 12(1), 2. Zhong, Y., Deng, L., Zhou, L., Liao, S., Yue, L., Wen, S.W., Xie, R., Lu, Y., Zhang, L., Tang, J. and Wu, J. 2022. Association of immediate reinsertion of new catheters with subsequent mortality among patients with suspected catheter infection: a cohort study. Ann. Intensive. Care. 12(1), 38. | ||
| How to Cite this Article |
| Pubmed Style zhu J, Xu Y, SW, FW, WZ, Wang R. Influencing factors of central venous catheter-associated bloodstream infection in patients with emergency hemorrhagic shock. J Microbiol Infect Dis. 2023; 13(3): 139-147. doi:10.5455/JMID.2023.v13.i3.6 Web Style zhu J, Xu Y, SW, FW, WZ, Wang R. Influencing factors of central venous catheter-associated bloodstream infection in patients with emergency hemorrhagic shock. https://www.jmidonline.org/?mno=152205 [Access: May 13, 2024]. doi:10.5455/JMID.2023.v13.i3.6 AMA (American Medical Association) Style zhu J, Xu Y, SW, FW, WZ, Wang R. Influencing factors of central venous catheter-associated bloodstream infection in patients with emergency hemorrhagic shock. J Microbiol Infect Dis. 2023; 13(3): 139-147. doi:10.5455/JMID.2023.v13.i3.6 Vancouver/ICMJE Style zhu J, Xu Y, SW, FW, WZ, Wang R. Influencing factors of central venous catheter-associated bloodstream infection in patients with emergency hemorrhagic shock. J Microbiol Infect Dis. (2023), [cited May 13, 2024]; 13(3): 139-147. doi:10.5455/JMID.2023.v13.i3.6 Harvard Style zhu, J., Xu, . Y., , . S. W., , . F. W., , . W. Z. & Wang, . R. (2023) Influencing factors of central venous catheter-associated bloodstream infection in patients with emergency hemorrhagic shock. J Microbiol Infect Dis, 13 (3), 139-147. doi:10.5455/JMID.2023.v13.i3.6 Turabian Style zhu, Jianping, Yanxin Xu, Shaohong Wu, Feiyao Wang, Weixing Zhang, and Ruilan Wang. 2023. Influencing factors of central venous catheter-associated bloodstream infection in patients with emergency hemorrhagic shock. Journal of Microbiology and Infectious Diseases, 13 (3), 139-147. doi:10.5455/JMID.2023.v13.i3.6 Chicago Style zhu, Jianping, Yanxin Xu, Shaohong Wu, Feiyao Wang, Weixing Zhang, and Ruilan Wang. "Influencing factors of central venous catheter-associated bloodstream infection in patients with emergency hemorrhagic shock." Journal of Microbiology and Infectious Diseases 13 (2023), 139-147. doi:10.5455/JMID.2023.v13.i3.6 MLA (The Modern Language Association) Style zhu, Jianping, Yanxin Xu, Shaohong Wu, Feiyao Wang, Weixing Zhang, and Ruilan Wang. "Influencing factors of central venous catheter-associated bloodstream infection in patients with emergency hemorrhagic shock." Journal of Microbiology and Infectious Diseases 13.3 (2023), 139-147. Print. doi:10.5455/JMID.2023.v13.i3.6 APA (American Psychological Association) Style zhu, J., Xu, . Y., , . S. W., , . F. W., , . W. Z. & Wang, . R. (2023) Influencing factors of central venous catheter-associated bloodstream infection in patients with emergency hemorrhagic shock. Journal of Microbiology and Infectious Diseases, 13 (3), 139-147. doi:10.5455/JMID.2023.v13.i3.6 |